DOC_ANK_TNKS_1
| Accession: | |
|---|---|
| Functional site class: | Tankyrase-binding motif |
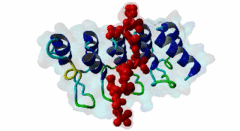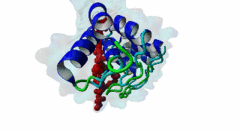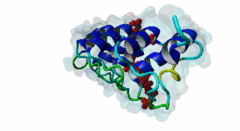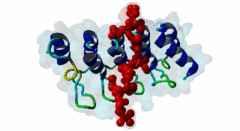
| Functional site description: | Tankyrases belong to the poly (ADP-ribose) polymerase (PARP) family of proteins, which function by catalyzing the covalent linkage of ADP-ribose polymers onto target proteins and thereby regulating their ubiquitylation, stability and function. The human genome encodes two similar tankyrases. TNKS and TNKS2 both recruit a variety of substrates with a broad range of functions. Each shows some variation in their conservation and tissue expression. Moreover both Tankyrases contain N-terminal ankyrin repeats. These ankyrin repeats form five domains known as ankyrin repeat clusters (ARCs) and each can serve as a discrete binding site for its binding partners. Tankyrase plays important roles in many biological functions including telomere length regulation, insulin signalling and centrosome function. Dysregulation of 3BP2 recognition by tankyrase causes a human disease knows as cherubism. Tankyrase is also considered to be a potential candidate for a telomere-directed anticancer target. |
| ELM Description: | Tankyrase1 and Tankyrase 2 are closely related poly (ADP-ribose) polymerases. They have both enzymatic and scaffolding activities. Tankyrases are multidomain proteins. The C-terminal PARP domain catalyzes the poly-(ADP-ribosyl)ation of substrate proteins, whereas the ankyrin repeat domain interacts with the substrates and brings them to PARP domain. There are five ankyrin triple repeat clusters (ARCs) present in Tankyrase and, except ARC3, all are predicted to act as independent binding unit. The ARCs interact with substrates through their tankyrase binding motifs. All ARCs having similar substrate recognition abilities and the overall binding mode of substrates to an ARC of Tankyrase is highly conserved, with the binding peptide lying in a surface groove. Each ARC is able to bind the same set of proteins with different binding affinity. So they are redundant in function. The Tankyrase binding motif is usually depicted as RxxPDG. The arginine and glycine residues at positions 1 and 6 are highly conserved acting as critical anchor residues of the motif. The R enters the deepest part of the groove making charged interactions. The G is sandwiched in the narrowest part of the groove. The P and D positions are more variable. The motif is experimentally verified in nearly 17 proteins including 3BP2, AXIN1, TRF1, IRAP and NUMA1. |
| Pattern: | .R..[PGAV][DEIP]G. |
| Pattern Probability: | 0.0003538 |
| Present in taxon: | Metazoa |
| Interaction Domain: |
Ank (PF00023)
Ankyrin repeat
(Stochiometry: 4 : 1)
|
Tankyrases are members of the poly (ADP-ribose) polymerases (PARPs). PARPs catalyze the linkage of ADP-ribose onto their substrates either as monomers or polymers using NAD+ as a source. Tankyrase is found in both the cytosol and nuclear compartments and is implicated in many important cellular functions ranging from telomere homeostasis and mitosis to vesicle trafficking (Zhang,2011). In mammals, the tankyrase protein family consists of tankyrase 1 and 2, which share 85% amino acid sequence identity. Tankyrases seem only be present in the Metazoa (multicellular animals). The tissue expression of the tankyrases varies; TNKS is relatively abundant in reproductive tissues whereas the TNKS2 is more ubiquitous. Tankyrase 1 contains four distinct domains. The N-terminus contains the natively disordered HPS domain, followed by a long ankyrin repeat region. This region is clustered into five well conserved subdomains of three ANK repeats each, designated as ARC (ANK repeat cluster) 1-V. Each ARC domain is able to act as an independent unit and can interact with a variety of substrates possessing the sequence region known as the tankyrase binding motif. The SAM (sterile alpha motif) domain contributes to the multimerization of tankyrase 1. The C-terminal region encompasses the PARP domain, which catalyses poly-ADP-ribosylation of acceptor proteins using NAD+ as a substrate (Seimiya,2006). Tankyrase 2 has a similar modular organization as tankyrase1 but lacks the natively disordered HPS domain. Both tankyrases use the ankyrin modules to dock their substrates via their tankyrase binding motif. This motif consists of a minimum of six consecutive amino acids RxxPDG, where only the R and G are always conserved (Guettler,2011). Protein PARsylation can affect the fate of the protein through several ways including direct modulation of its activity, recruitment of binding partners that recognize poly(ADP-ribose) or by affecting protein turn over. Recent studies have shown that the mutation in the TNKS-binding motif of 3BP2 disrupts the interaction with Tankyrase and leads to cherubism in humans. |
-
Identification of a tankyrase-binding motif shared by IRAP, TAB182, and
human TRF1 but not mouse TRF1. NuMA contains this RXXPDG motif and is a
novel tankyrase partner.
Sbodio JI, Chi NW
J Biol Chem 2002 Aug 30; 277 (35), 31887-92
PMID: 12080061
-
Functional subdomain in the ankyrin domain of tankyrase 1 required for
poly(ADP-ribosyl)ation of TRF1 and telomere elongation.
Seimiya H, Muramatsu Y, Smith S, Tsuruo T
Mol Cell Biol 2004 Mar; 24 (5), 1944-55
PMID: 14966275
-
The telomeric PARP, tankyrases, as targets for cancer therapy.
Seimiya H
Br J Cancer 2006 Feb 13; 94 (3), 341-5
PMID: 16421589
-
Tankyrase function at telomeres, spindle poles, and beyond.
Hsiao SJ, Smith S
Biochimie 2008 Jan; 90 (1), 83-92
PMID: 17825467
-
RNF146 is a poly(ADP-ribose)-directed E3 ligase that regulates axin
degradation and Wnt signalling.
Zhang Y, Liu S, Mickanin C, Feng Y, Charlat O, Michaud GA, Schirle M, Shi X, Hild M, Bauer A, Myer VE, Finan PM, Porter JA, Huang SM, Cong F
Nat Cell Biol 2011 May; 13 (5), 623-9
PMID: 21478859
-
Structural basis and sequence rules for substrate recognition by Tankyrase
explain the basis for cherubism disease.
Guettler S, LaRose J, Petsalaki E, Gish G, Scotter A, Pawson T, Rottapel R, Sicheri F
Cell 2011 Dec 9; 147 (6), 1340-54
PMID: 22153077
(click table headers for sorting; Notes column: =Number of Switches, =Number of Interactions)
Please cite:
ELM-the Eukaryotic Linear Motif resource-2024 update.
(PMID:37962385)
ELM data can be downloaded & distributed for non-commercial use according to the ELM Software License Agreement
ELM data can be downloaded & distributed for non-commercial use according to the ELM Software License Agreement